 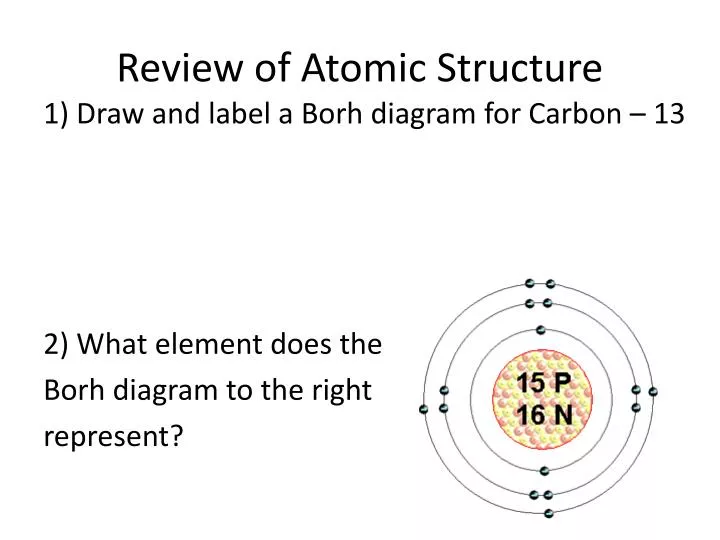
size, mass, density, chemical properties, but they differ from the atoms of other elements. Atoms can neither be created nor be destroyed The atoms of an element are identical in all respects i.e. Protons and neutrons each are given an amu of 1. John Daltons atomic theory or model Matter consists of very small and indivisible particles called atoms. A cathode ray tube is made of glass containing two thin pieces of metal called electrodes, sealed in it. The stages in this process are Atomic Structure. Structure of Atom 1 GRADE 11 DISCOVERY OF AN ELECTRON An electron was discovered by cathode ray discharge tubes experiment. Atomic mass is recorded in the SI units: atomic mass units (amu). Presenting this set of slides with name - Atomic Structure Template Ppt Icon. The crystal is cut parallel to a set of its -planes and bombardment is at normal incidence. Atom Characteristics The atomic mass is the mass of the protons plus the mass of the neutrons. it" of scattering of a homogeneous beam of electrons of adjustable speed incident upon a single crystal of nickel has been measured as a function of direction.Germer experiment Electron scattering peak at 500 1929 Nobel Prize for de Broglie PHYSICAL REVIEW DIFFRACTION OF ELECTRONS BY A CRYSTAL OF NICKEL BY C. Since the iodine is added as a 1 anion, the number of electrons is 54. Because the sum of the numbers of protons and neutrons equals the mass number, 127, the number of neutrons is 74 (127 53 74). 30, 6 1924 de Broglie's hypothesis 0 Nickel stal 1927 Davisson. The atomic number of iodine (53) tells us that a neutral iodine atom contains 53 protons in its nucleus and 53 electrons outside its nucleus. Electron gun hot filament to Mrelease electrons +54 V Accelerating o electrode Theory -1.67Åfor54V Experiment Pathlength difference d sine 1.65Å for constructive interference Not bad for a three ear old idea! Nickel lattice spacing d = 2,15Å Second Series December, 1927 THE Vol. Atomic Structure Slide 3 / 100 Chemistry Chemistry is the study of matter and the changes it undergoes. They discovered the wave nature of electron by getting a diffraction pattern from a Ni crystal by bombarding a beam of electron. All elements are composed of tiny indivisible particles called atoms. Experimental proof of de Broglie hypothesis Davisson-Germer experiment (1923-1927) confirms the de Broglie hypothesis (wave-particle duality). 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
